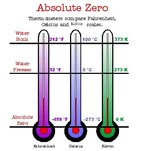
Absolute Zero: the lowest temperature that is theoretically possible, at which the motion of particles that constitutes heat would be minimal.
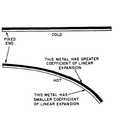
Bimetallic Strip: a temperature-sensitive electrical contact used in some thermostats, consisting of two bands of different metals joined face to face along their lengths. When heated, the metals expand at different rates, causing the strip to bend.

Calorie: either of two units of heat energy

Celsius Scale: the scale of temperature in which water freezes at 0° and boils at 100° under standard conditions
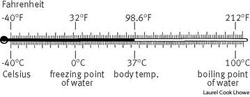
Fahrenheit Scale: the Fahrenheit scale of temperature.

Heat: the quality of being hot; high temperature
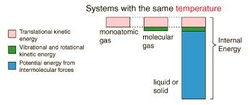
Internal Energy: the energy in a system arising from the relative positions and interactions of its parts.

Kelvin Scale: a scale of temperature with absolute zero as zero, and the triple point of water as exactly 273.16 degrees

Kilocalorie: a unit of energy of 1,000 calories (equal to 1 large calorie)
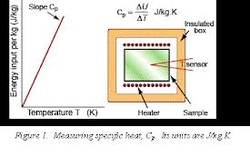
Specific Heat Capacity:the heat required to raise the temperature of the unit mass of a given substance by a given amount (usually one degree)
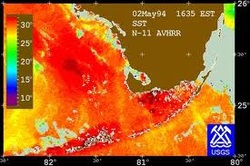
Temperature: the degree or intensity of heat present in a substance or object, esp. as expressed according to a comparative scale and shown by a thermometer or perceived by touch.
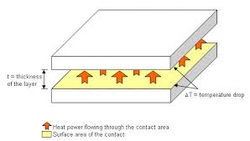
Thermal Contact: In heat transfer and thermodynamics, a thermodynamic system is said to be in thermal contact with another system if it can exchange energy with it through the process of heat.
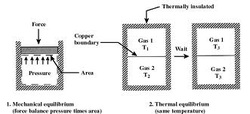
Thermal Equilibrium: a state in which all parts of a system are at the same temperature

Thermostat: a device that automatically regulates temperature, or that activates a device when the temperature reaches a certain point.
